Solubility PVP polymer is readily soluble in cold water and the concentration is limited only by viscosity. These are most often phosphate ammonium or carboxylate all of which are charged when dissolved in an aqueous solution buffered to pH 7.
1
1 6 Physical Properties Of Organic Compounds Chemistry Libretexts
Ternary Phase Equilibrium Data For Acetic Acid Water Solvent Systems And Separation Of Acetic Acid From Aqueous Solution Scialert Responsive Version
Properties of Organic Solvents.

Butanol solubility in water. C 28 H 31 ClN 2 O 3. 27 EPI Suite 28. When alkyl group gets longer or larger solubility of alcohols decreases.
Because water is the biological solvent most biological organic molecules in order to maintain water-solubility contain one or more charged functional groups. 2-Butanol ist das einzige Butanol mit einem Chiralitätszentrum so dass zwei optisch aktive Enantiomere vorkommen. The values in the table below except as noted have been extracted from online and hardbound compilations.
RhB is a fluorescent dye with good solubility in water and high stability to light chemical formula. Further the effect of pH variation on the solubilities of the amino acids in each of the solvent systems was studied. 081 20C4C pH.
11 The Nature of the Project. Solubility in water is limited and decreases with increasing 11 temperature. Remove contaminated clothing and shoes.
Water is the single most abundant and important liquid on this planet. Solubility of these lipids increase in alcoholic solvents as the carbon chain length of the alcohol increases so they are more soluble in ethanol and n-butanol. Butyl alcohol was probably partially taken up in the water of the dead space mucous membranes.
Propanol dissolve little bit. Natürlich kommt 2-Butanol in Äpfeln Malus domestica Schwarzen Johannisbeeren Ribes nigrum und Myrte Myrtus communis vor. 12 2-MTHF is increasingly used as a catalytic solvent in exchange of Tetrahydrofuran THF and is 13 much less miscible with water compared to THF.
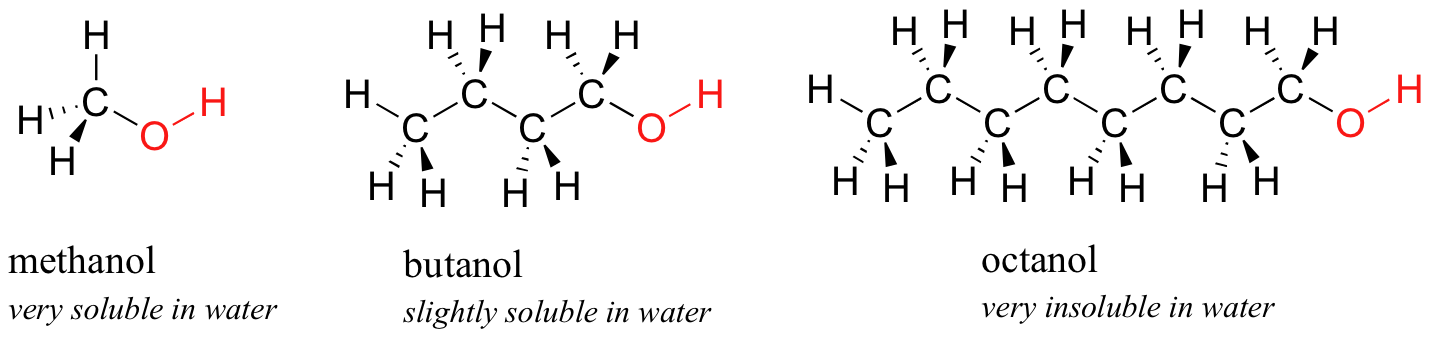
The higher alcoholsthose containing 4 to 10 carbon atomsare somewhat viscous or oily and they have heavier fruity odours. Octanol water partition coefficient is typically expressed in logarithm form and is indicated as log K ow or log PThis parameter is very common and it is obtained using experimental procedures 2224 using estimation techniques 25 26 or it is evaluated using computer programs based on models eg MarvinSketch 5401 ChemAxon Ltd. Starting with the four-carbon butanol the solubility of alcohols is starting to decrease.
1-Butanol Spectranalyzed Revision Date 08-May-2019 General Advice If symptoms persist call a physician. Tert-Butyl alcohol is the simplest tertiary alcohol with a formula of CH 3 3 COH sometimes represented as t-BuOHIt is one of the four isomers of butanol. Methanol ethanol dissolve very well in water.
10 enantiomeric forms S and R-. Introduction to the Solubility Data Series. The quotient of alveolar concentration X 100inspired concentration was low in relation to the low percentage uptake.
Aliquots were withdrawn from each solvent system studied and analyzed gravimetrically to determine the resultant solubility. Immediately flush skin with plenty of water for at least 15 minutes. Because of the strength of the attraction of the OH group first three alcohols methanol ethanol and propanol are completely miscible.
N-butanol and isobutanol have limited solubility sec-butanol has substantially greater solubility while tert-butanol is miscible with water. Eye Contact Rinse immediately with plenty of water also under the eyelids for at least 15 minutes. The Solubility Data Project SDP has as its aim a comprehensive review of published data for solubilities of gases liquids and solids in liquids or solids.
Wenn in diesem Artikel oder in der wissenschaftlichen Literatur 2-Butanol ohne eine zusätzliche Angabe zur Konfiguration erwähnt. But due to large phenyl group phenol does not dissolve in water. Urine was analyzed immediately 1 2 8 and 9 hr after drinking during 2 hr 375 mlkg of beverages containing orange juice 15 or 40 ethanol and 1 gl of 1-propanol 2-propanol 1-butanol 2-butanol isobutyl alcohol or a mixture of 1-propanol isobutyl alcohol.
Skin Contact Wash off immediately with plenty of water for at least 15 minutes. Values for relative polarity eluant strength threshold limits and vapor pressure have been extracted from. It was found that the solubility.
Alcohol - alcohol - Physical properties of alcohols. The butanol isomers have different melting and boiling points. IUPAC-NIST Solubility Data Series.
Water isopropanol-water and tertiary butanol-water. It is possible to prepare free-flowing solutions of PVP K-30 polymer in concentrations up to 60 with only moderate effect on density. PVP K-60 and K-90 polymer are available commercially as 45 and 20 percent aqueous solutions respectively.
Christian Reichardt Solvents and Solvent Effects in Organic Chemistry Wiley-VCH Publishers 3rd ed 2003. The miscibility of other liquids in water and the solubility of solids in water must be considered when isolating and purifying compounds. Distilled water Russian standard GOST 6709-2 was mixed with two typical fluorophores dyes Rhodamine B RhB and Rhodamine 6G Rh6G in turn.
It has a vapour pressure of 136 mbar 20C 1. Page 1 of 7 MSDS N-Butanol Material Safety Data Sheet MSDS- N-BUTANOL 1. The shorter chain fatty acids in the lipids will have greater solubility in the more polar solvents.
Methyl alcohol ethyl alcohol and isopropyl alcohol are free-flowing liquids with fruity odours. Solubility of alcohols is therefore determined by the stronger of the two forces. Most of the common alcohols are colourless liquids at room temperature.
Butanol is completely insoluble in water. However the high solubility of butyl alcohol in water may explain the results. They dissolve in water in any amount.
Tert-Butyl alcohol is a colorless solid which melts near room temperature and has a camphor-like odorIt is miscible with water ethanol and diethyl ether. The hydroxyl group makes the molecule polar promoting solubility in water while the longer hydrocarbon chain mitigates the polarity and reduces solubility. Phenol can make hydrogen bonds due to -OH bond.
9 mL100 mL water 25C Specific Gravity. Rh6G is a fluorescent dye used in luminescence microscopy. If skin irritation persists call a physician.
To this end the following table lists the water miscibility or solubility of an assortment of low molecular.
Degradation Of Tert Butyl Alcohol In Water At Ph 3 0 Tba 0 1 629 G L Download Scientific Diagram

Solubility Of Water In 1 Butanol Gasoline Blends Expressed As The Download Scientific Diagram

1 Butanol Wikipedia
Sciencedirect Com

Solubility Of Water In 1 Butanol Gasoline Blends Expressed As The Download Scientific Diagram
Solved Solubility Behavior Indicate If The Sample Was Chegg Com

12 35 The Solubility Of N Butanol In Water Is 7 7 Chegg Com

Tert Butyl Alcohol Wikipedia

