They also have a positive influence on the quality of the result. The purpose of the.

Introduction Of Good Laboratory Practice Glp Tower Mains

Good Laboratory Practice Regulations Introduction And Strategies For Implementation Youtube

An Introduction To Good Laboratory Practices 01 Tobin Emmet Amazon Com
How to get the most from the course.
Good laboratory practice. The principles have been developed in accordance with the Organisation for Economic Cooperation and Development. EPAs Good Laboratory Practice Standards GLPS compliance monitoring program ensures the quality and integrity of test data submitted to the Agency in support of a pesticide product registration under the Federal Insecticide Fungicide and Rodenticide Act FIFRA section 5 of the Toxic Substances Control Act TSCA and pursuant to testing consent agreements and test rules issued under. In the experimental non-clinical research arena good laboratory practice or GLP is a quality system of management controls for research laboratories and organizations to ensure the uniformity consistency reliability reproducibility quality and integrity of products in development for human or animal health including pharmaceuticals through non-clinical safety tests.
From physio-chemical properties through acute to. Good laboratory practice or good laboratory practices are accepted methods to carry out activities or operations in a laboratory. The pertinent regulations in the preclinical scenario are the Good Laboratory Practice GLP regulations.
The Food and Drug Administration FDA is proposing to amend the regulations for good laboratory practice GLP for nonclinical laboratory studies to require a complete quality system approach referred to as a GLP Quality System when safety and toxicity studies support or. Introduction to Good Clinical Laboratory Practice is a stand-alone short course for all those wanting to gain an understanding of GCLP and its application in laboratories. The principles of Good Clinical Practice GCP help assure the safety integrity and quality of clinical trials by addressing elements related to the design conduct and reporting of clinical trials.
These regulations are the subject of this handbook which is a reference and support document to help in the implementation of GLP. The principles of Good Laboratory Practice GLP define a set of rules and criteria for a quality system concerned with the organisational process and the conditions under which non-clinical health and environmental safety studies are planned performed monitored recorded reported and archived. Data and research on test guidelines including chemical testing and assessment chemical safety animal welfare endocrine disrupters good laboratory practice GLP Mutual Acceptance of Data MAD Read the countries response to address the challenges posed by COVID-19 to GLP test facilities.
The authorities and laboratory organizations say that these practices help ensure safety. The principles of Good Laboratory Practice GLP promote the quality and validity of data generated in the testing of chemicals and prevent fraudulent practices. The OECD Principles of Good Laboratory Practice OECD Series on Principles of Good Laboratory Practice and Compliance Monitoring Number 1 ENVMCCHEM9817 or subsequent revisions are adopted as the GLP standard by the Pest Management Regulatory Agency PMRA.
We describe eleven core elements that constitute the. The purpose of testing these items is to obtain information on their safety with respect to human health and environment. Food and Drug Administration.
Subpart G - Protocol for and Conduct of a Nonclinical Laboratory Study 58120 - 58130 Subparts H-I Reserved Subpart J - Records and Reports 58185 - 58195 Subpart K - Disqualification of Testing Facilities 58200 - 58219. Any test facility which conducts or intends to conduct regulatory studies must comply with good laboratory practice GLP regulations when carrying out safety tests onpharmaceuticals. GLP Good laboratory practices Good laboratory practice or GLP specifically refers to a quality system of management controls for research laboratories and organizations to try to ensure the uniformity consistency reliability reproducibility quality and integrity of chemical including pharmaceuticals non- clinical safety tests.
Good Laboratory Practice GLP are federal regulations that require implementation of a robust quality management system to ensure the validity integrity and reliability of non-clinical safety data submitted for regulatory evaluation and approval. A set of Good Clinical Laboratory Practice GCLP standards that embraces both the research and clinical aspects of GLP were developed utilizing a variety of collected regulatory and guidance material. GCP training describes the responsibilities of investigators sponsors monitors and.
Good Laboratory Practice GLP is defined in the Organisation for Economic Co-operation and Development OECD as a quality system concerned with the organisational process and the conditions under which non-clinical health and environmental safety studies are planned performed monitored recorded archived and reported. This is the complete set of the series on OECD Principles of Good Laboratory Practice GLP which set the quality standards for the organisation and management of test facilities and for performing and reporting studies related to the safety of chemical substances and preparations. Good Clinical Laboratory Practice is a multi-module course for all those wanting to gain an in-depth understanding of GCLP and how each principle of GCLP can be applied in clinical trial laboratory.
The most well-known is The Good Automated Manufacturing Practice GAMP Guide for Validation of Automated Systems in Pharmaceutical ManufactureThe last major revision GAMP5 was released in February 2008. Data and research on test guidelines including chemical testing and assessment chemical safety animal welfare endocrine disrupters good laboratory practice GLP Mutual Acceptance of Data MAD This OECD Advisory Document on Good Laboratory Practice GLP Data Integrity provides guidance for test facilities or test sites that conduct GLP studies and aims to promote a risk-based approach. Good laboratory practice compliance This content applies to human and veterinary medicines.
ISPE has published a series of good practice guides for the industry on several topics involved in drug manufacturing. Good Laboratory Practice GLP is a quality system concerned with the organisational process and the conditions under which non-clinical health and environmental safety studies are planned. This peer reviewed course provides an introduction to GCLP summarises the principles of GCLP and offers an overview of the implementation of GCLP within a clinical trial.
Good Laboratory Practice Regulations 1981 GLP Questions Answers SUBPART A GENERAL PROVISIONS Section 581 - Scope. The Principles of Good Laboratory Practice of the Organisation for Economic Cooperation and Development. Do the GLPs apply to validation trials conducted to.
Good Laboratory Practice GLP regulations are applied to non-clinical safety of study items contained in pharmaceutical products cosmetic products veterinary drugs devices as well as food additives.

Glp Online Training Course Good Laboratory Practice

Eposters Get Good Laboratory Practice Certified
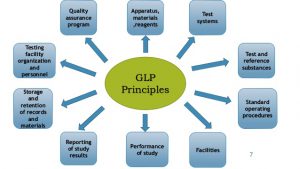
Glp Good Laboratory Practice System New Version 2021

General Rules For Laboratories Operating Under Good Laboratory Practice Glp

Good Laboratory Practice

Accutest Biologics Bags Oecd Good Laboratory Practice Certification Express Pharma

What Is Good Laboratory Practice Glp Definition And Meaning
83 Good Laboratory Practices Illustrations Clip Art Istock
